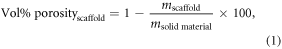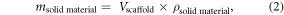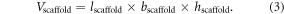Abstract
Bottom-up biofabrication approaches combining micro-tissue fabrication techniques with extrusion-based 3D printing of thermoplastic polymer scaffolds are emerging strategies in tissue engineering. These biofabrication strategies support native self-assembly mechanisms observed in developmental stages of tissue or organoid growth as well as promoting cell–cell interactions and cell differentiation capacity. Few technologies have been developed to automate the precise assembly of micro-tissues or tissue modules into structural scaffolds. We describe an automated 3D bioassembly platform capable of fabricating simple hybrid constructs via a two-step bottom-up bioassembly strategy, as well as complex hybrid hierarchical constructs via a multistep bottom-up bioassembly strategy. The bioassembly system consisted of a fluidic-based singularisation and injection module incorporated into a commercial 3D bioprinter. The singularisation module delivers individual micro-tissues to an injection module, for insertion into precise locations within a 3D plotted scaffold. To demonstrate applicability for cartilage tissue engineering, human chondrocytes were isolated and micro-tissues of 1 mm diameter were generated utilising a high throughput 96-well plate format. Micro-tissues were singularised with an efficiency of 96.0 ± 5.1%. There was no significant difference in size, shape or viability of micro-tissues before and after automated singularisation and injection. A layer-by-layer approach or aforementioned bottom-up bioassembly strategy was employed to fabricate a bilayered construct by alternatively 3D plotting a thermoplastic (PEGT/PBT) polymer scaffold and inserting pre-differentiated chondrogenic micro-tissues or cell-laden gelatin-based (GelMA) hydrogel micro-spheres, both formed via high-throughput fabrication techniques. No significant difference in viability between the construct assembled utilising the automated bioassembly system and manually assembled construct was observed. Bioassembly of pre-differentiated micro-tissues as well as chondrocyte-laden hydrogel micro-spheres demonstrated the flexibility of the platform while supporting tissue fusion, long-term cell viability, and deposition of cartilage-specific extracellular matrix proteins. This technology provides an automated and scalable pathway for bioassembly of both simple and complex 3D tissue constructs of clinically relevant shape and size, with demonstrated capability to facilitate direct spatial organisation and hierarchical 3D assembly of micro-tissue modules, ranging from biomaterial free cell pellets to cell-laden hydrogel formulations.

Original content from this work may be used under the terms of the Creative Commons Attribution 3.0 licence. Any further distribution of this work must maintain attribution to the author(s) and the title of the work, journal citation and DOI.
1. Introduction
In tissue engineering and regenerative medicine, conventional 'top-down' fabrication strategies revolve around populating cells within a 3D scaffold [1]. With regards to cartilage tissue engineering, the typical paradigm involves in growing chondrocytes or chondroprogenitor cells in combination with cell phenotype modulating growth factors within a biodegradable 3D scaffold in vitro, and then implanting the engineered construct into the target site [2]. This approach offers advantages by providing the initial mechanical properties, cells and extracellular matrix (ECM) components required to elicit regeneration and tissue repair [3]. Although significant advances have been made in tissue engineering, major challenges are yet to be conquered before cartilage tissue engineering strategies can be applied in a clinical setting [4]. For example, chondrocytes need to be guided to prevent dedifferentiation and promote the generation of high quality repair tissue in vivo that mimics the complex cellular and ECM organisation in native hyaline cartilage, and thereby allow normal load bearing and long-term function of the joint [5].
Though the use of top-down biofabrication techniques for producing scaffolds have come of age [6], recreating the complex microarchitecture of native tissue utilising top-down approaches is challenging [1]. An alternative approach to addressing these challenges is by adopting 'bottom-up' strategies, where smaller nano or microscale tissue units are assembled to create a larger more complex engineered tissue construct with improved control over architecture [7]. With respect to bottom-up approaches, Livoti and Morgan et al [8, 9] reported on a tissue fusion technique, whereby a printing approach was used to produce complex shapes that were assembled using micro-tissues as building units on agarose moulds. Jakab et al [10] demonstrated a system for the self-assembly of micro-tissues using a bioprinter device that cuts and extrudes cellular slurry into cylindrical aggregates for delivery within a hydrogel substrate to promote tissue fusion and maturation. In addition, a spectra of techniques have been demonstrated to assemble prefabricated tissue modules into higher order tissue structures. These techniques include: stacking of cell sheets [11], random packing of microgels [12], utilising hydrophobic effects for assembling microgels [13], physical manipulation of individual cell-laden microgels [14], magnetically driven assembly of microgels [15, 16], acoustic wave driven assembly of microgels [17], assembly of microgels with micro-robotic systems [18], vacuum moulding [19, 20], stacking rings on a tube [21], assembling micro-tissues by stacking them on skewers [22], or manual assembly of micro-tissues within 3D plotted scaffolds [23].
Tissue assembly technologies with the ability to precisely place spherical micro-tissues into specific locations within 3D plotted structural scaffolds for developing hybrid hierarchical constructs have been limited [6], and the manual assembly of micro-tissues has currently been confined to small, simple constructs. To our knowledge, there are few approaches describing the combination of automated bottom-up bioassembly approaches for hierarchical biofabrication and direct 3D bioassembly of micro-tissues in complex, anatomically-shaped 3D scaffolds.
Micro-mass or pellet culture of chondrocytes or chondroprogenitor cells are well-established methods for generating small micro-tissues of engineered cartilage in vitro, and more recent studies have investigated high throughput approaches for micro-tissue formation [23, 24]. High throughput generation of micro-tissues has been demonstrated with: (i) hanging drop technology [25]; (ii) 96-well plate formats for cartilage engineering [23], assessing chondrogenesis [26, 27] and for screening of anti-rheumatic compounds [28]; and (iii) a 384-well plate format for high throughput screening of chondrogenesis in mesenchymal stromal cells (MSC) [29]. A previous study by Schon et al [23], demonstrated a high throughput technique for producing ex vivo cartilage-like micro-tissues. In these micro-tissue, the 3D niche promotes large number of cell–cell interactions [30], thereby replicating the cellular condensation environment typical during developmental stages of cartilage growth, allowing the production of hyaline-like ex vivo neocartilage expressing collagen type II and aggrecan [23] typical of mature hyaline cartilage. Consequently, an opportunity exists for an automated approach for the high throughput culture and bioassembly of micro-tissues within a hybrid scaffold to obtain a functional tissue engineered construct consisting of large number of pre-differentiated micro-tissues primed for promoting ECM formation [31]. The size of the micro-tissue may further promote or restrict matrix formation, conditional to the resultant oxygen and nutrient diffusion gradients. Cartilage is an avascular tissue and it is recognised that a hypoxic environment stimulates chondrocyte differentiation and ECM production in vitro [32–34]. Hypoxic signalling (hypoxia-inducible transcription factors HIF-1 and HIF-2) plays an important role in oxygen homeostasis, is also known to affect chondrocyte survival and cartilage tissue formation [35, 36]. Subsequently, most literature investigating differentiation of small <300 μm high-throughput micro-tissues describe that hypoxic culture conditions are required to stimulate chondrogenic differentiation given that <300 μm spheroids do not have sufficient dimensions to support an inherent hypoxic gradient or core [37]. A large number of publications, including our own work, instead describe the high-throughput fabrication and successful long-term culture of micro-tissues ranging in diameter over time from 0.7 to 1.4 mm, without undifferentiated or necrotic cells present in the central regions evident by the deposition of cartilaginous matrix components [23, 30, 38–42, 67]. As the field of microscale tissue engineering expands, biofabrication of microscale tissue units or micro-spheres made of cell-laden hydrogels are further considered an attractive approach to gain more precise control and manipulation of micro-environmental factors [43–45]. Gelatin and Heparin based hydrogel formulations have herein been highlighted as inexpensive and cell-instructive hydrogel platforms compatible with emerging biofabrication technologies to help guide cellular development [44, 46–48]. Combined, these literature reports served as a basis for adopting 1 mm diameter spherical modular micro-tissues and micro-spheres as model assembly units in this study.
The absence of a roadmap for scalable, reproducible, automated, large-scale biofabrication of tissue engineered products with a high level of cell–cell interaction, cellular self-assembly and co-location in combination with structurally reinforcing scaffolds offers opportunities to develop modular bioassembly technologies [49]. The requirements of such a system would include: (i) high degree of automation and scalability, (ii) ability to assemble micro-tissues of different cell types with controlled or hierarchical spatial resolution (e.g. osteochondral biphasic constructs [50]), (iii) ability to fabricate large, complex and high fidelity constructs of clinically relevant size. The system must also be able to promote rapid cell differentiation and ECM formation that mimics the complex 3D architecture of the native tissue as well as reduce overall construct fabrication time.
For the advancement of biofabrication approaches, any 3D bioassembly technology should preferably offer flexibility and be highly capable of fabricating both simple and anatomically complex hybrid tissue constructs. We envisioned that the 3D bioassembly process should be capable of two specific bioassembly strategies: (i) a two-step bottom-up bioassembly process involving the prefabrication of a 3D plotted scaffold and the assembly of specific tissue units allowing biofabrication of simple hybrid constructs and (ii) a multistep bottom-up bioassembly strategy involving the layer-by-layer fabrication of 3D plotted scaffold and co-assembly of specific tissue units, enabling the biofabrication of complex hybrid constructs. Figure 1 illustrates the opportunities offered by advanced modular tissue bioassembly strategies for biofabrication of both multicellular 3D tissue models and complex anatomical hybrid constructs for high throughput screening and tissue engineering applications.
Figure 1. Illustration of an automated bottom-up strategy for 3D bioassembly of hybrid, hierarchical constructs. (a) A 3D design is translated from a computer aided model to a 3D plotted construct. (b) Simple two-step bottom-up bioassembly strategy: involving 3D plotting a scaffold and subsequent spatial assembly of single or multicellular micro-tissues and (b) complex multistep bottom-up bioassembly strategy: layer-by-layer assembly process alternating between 3D plotting and multicellular micro-tissue insertion allowing biofabrication of complex, anatomically shaped hybrid constructs.
Download figure:
Standard image High-resolution imageThe specific aim of this study was therefore to develop an automated biofabrication platform combining 3D micro-tissue bioassembly and 3D plotting of thermoplastic polymer scaffolds. This involved the development of a system consisting of (i) a singularisation module that is able to separate large numbers of engineered spheroids or micro-tissues and then deliver them individually to an injection module, (ii) an injection module which can assemble or seed the micro-tissue at a predetermined location within a defined scaffold architecture with the aid of a 3D positioning system. The ultimate goal of this technology is the development of an automated biofabrication system capable of assembling living constructs suitable for clinical translation, preliminarily targeting cartilage repair strategies.
2. Methods
2.1. Scaffold fabrication
Biodegradable poly(ethylene glycol)-terephthalate-poly(butylene terephthalate) block copolymers (Polyactive 300PEGT55PBT45, PolyVation, The Netherlands) with a PEG molecular weight (MW) of 300 g mol−1 and a PEGT:PBT weight percent (wt%) ratio of 55:45 were used to fabricate scaffolds with a specific pore size and architecture. PEGT/PBT copolymer composition was selected as a model scaffold based on previous studies demonstrating its applicability across multiple processing techniques (e.g. melt extrusion), mechanical properties mimicking native tissue, as well as its capacity to modulate cell, adhesion, proliferation, differentiation and extracellular matrix formation [2, 23, 51–55]. Porous scaffolds (15 × 15 × 3.5 mm), with accurately defined and controlled pore architecture for micro-tissue incorporation, were 3D plotted using a BioScaffolder system (SYS ENG, Germany). Fibres were oriented in a repeating 0–90°–90°–0° pattern in order to provide porosity in both the x–y and x–z planes for assembly of 1 mm diameter micro-tissues (figure 2). During the melt dispensing process the following 3D plotting parameters were applied: (i) fibre spacing of 1 mm in both x and y direction, (ii) fibre height offset of 0.22 mm, (iii) dispense head and plotting temperature of 200° and 5 bar pressure, (iv) dispense head auger speed of 63 RPM and (v) 25 gauge dispense head nozzle moving at a x–y traverse speed of 500 mm min−1. In the z-plane, the number of layers of 3D plotted fibres per layer of micro-tissue was such that they were high enough to accommodate a micro-tissue (figure 2(b)).
Figure 2. (a) Top view and (b) sectional front view of the specifically designed 3D plotted scaffold for micro-tissue bioassembly. The dotted circle denotes the position of the bioassembled micro-tissues within the scaffold pore volume.
Download figure:
Standard image High-resolution image2.2. Scaffold characterisation
3D plotted scaffold architectures designed for bioassembly were sputter coated with gold (Emitech K957X), after which scanning electron microscopy (SEM) images were taken using a Jeol 7000F FE-SEM with secondary electron detection used at an acceleration voltage of 15 kV. The interconnecting pore size of the 3D plotted scaffolds in the x–y plane and z plane and the fibre diameter was measured from calibrated SEM images. Rendered images illustrating the bioassembly scaffold design, interconnecting pore size and fibre diameter measurements are shown in figure 2.
The volume percentage porosity (Vol% porosityscaffold) of the 3D plotted scaffold was determined by mass-volume method using equation (1) as described previously [2]. The length (lscaffold), breath (bscaffold), height (hscaffold) and the mass (mscaffold) of the 3D plotted scaffold was measured.
where msolid material was determined using equations (2) and (3) and the density of polyactive 300PEGT55PBT45 (ρsolid material = 1.25 g cm−3) was based on the value previously reported and the theoretical value of porosity was also determined as described earlier [2].
The unconfined dynamic stiffness (compressive modulus) of the 3D plotted scaffolds was measured using an MTS Criterion® 42 mechanical test machine incorporating a 500 N load cell. 3D plotted scaffolds with a dimension of 5 × 5 × 1.8 mm were tested at room temperature under dry conditions and a preload of 0.1 N was applied to ensure that the compression plate was in contact with the sample. The dynamic stiffness was determined by applying a uniaxial unconfined cyclic compression between 1% and 5% strain (linear region of the stress–strain curve) at a frequency of 1 Hz. During cyclic compression, the equilibrium peak force was reached after approximately 70 cycles out of a total of 100 compression cycles. The dynamic stiffness was calculated by taking the average modulus for the last 10 cycles.
2.3. Cartilage excision and cell isolation
Human chondrocytes were isolated, expanded and formed into micro-tissues as described previously [23, 30]. Briefly, nasal or articular cartilage biopsies were obtained following ethics approval from consenting patients undergoing septoplasty surgery or cruciate ligament reconstruction, respectively (n = 2; 18, 25 years of age). The cartilage was diced into 1 mm cubes and digested overnight in basic chondrocyte media containing DMEM (high glucose, GlutaMAX Supplement, pyruvate; Gibco, USA) with 5% fetal bovine serum (FBS; Gibco, New Zealand), 0.1 mM non-essential amino acids (NEAA; Gibco, USA), 10 mM HEPES (Gibco, Taiwan), 0.4 mM L-proline (Sigma-Aldrich, USA), 100 units ml−1 penicillin (Gibco, USA) and 100 μg ml−1 streptomycin (Gibco, USA) containing 0.15% (w/v) collagenase type II (Worthington, USA). The suspension was then filtered through a 100 μm cell strainer (Corning, USA) and centrifuged at 700 g for 4 min.
2.4. Cell expansion
Freshly isolated human articular chondrocytes (HACs) or human nasal chondrocytes (HNCs) were seeded at a density of 3000 cells cm−2 in tissue culture flasks (Corning, USA) in basic chondrocyte media. Cells were expanded at 37 °C in a humidified 5% CO2/95% air incubator and media changed twice a week. After approximately 7 days, subconfluent cells were washed with phosphate-buffered saline (PBS; Gibco, USA), detached using 0.25% trypsin/EDTA (Gibco, Canada), counted by trypan blue exclusion in a haemocytometer and plated in a tissue culture flask at 3000 cells cm−2. Passage 2 (P2) cells were harvested similarly and utilised to form micro-tissues.
2.5. High throughput micro-tissue fabrication
Spherical micro-tissues were fabricated using a simple, high-throughput 96-well plate format previously developed in our group [23], which allows precise control and repeatability over micro-tissue size and shape. Briefly, micro-tissues—each consisting of 0.25 × 106 chondrocytes—were cultured in serum free chondrogenic differentiation media supplemented with 1% ITS+ (1 mg ml−1 insulin from bovine pancreas, 0.55 mg ml−1 human transferrin, 0.5 μg ml−1 sodium selenite, 50 mg ml−1 bovine serum albumin and 470 μg ml−1 linoleic acid; Sigma), 0.1 × 10−6 M dexamethasone (Sigma, USA), 1.25 mg ml−1 bovine serum albumin (Gibco, New Zealand) and 10 ng ml−1 recombinant human transforming growth factor-β1 (TGF-β1; R&D systems, USA). Cell suspensions consisting of 0.25 × 106 chondrocytes in 290 μl media were pipetted into each well of a polypropylene 96-well V-bottom plate (Raylab, New Zealand), and centrifuged (Eppendorf 5810) at 200 g for 4 min and placed in an incubator at 37 °C, 5% CO2. The following day, the newly formed micro-tissue was gently detached from the bottom of the V-plate by pipetting. During the subsequent micro-tissue culture period the media was changed 3 times per week and samples harvested on day 7.
2.6. Long-term culture and fusion of micro-tissues
A bioassembly fusion model system was designed to investigate the integration of micro-tissues over time as the possible formation and fusion of bioassembled tissue units may limit oxygen and nutrient diffusion and ultimately the functionality of the tissue engineered construct. Spherical micro-tissues were fabricated as described above and 3D plotted PEGT/PBT scaffolds composed of six layers oriented 0, 90, 0, 0, 90, 0 were fabricated to hold micro-tissues in place. Fibre spacing was 1 mm in the y direction and 3 mm in the x direction. Two micro-tissues were precisely bioassembled into the desired location within the scaffold and cultured in 96-well plates for up to 3 weeks in chondrogenic differentiation media as described above.
2.7. Design of the singularisation system
The singularisation device is a subsystem of the integrated tissue assembly device that collects the pooled micro-tissues from the high throughput fabrication process into a reservoir hopper, and then delivers a single micro-tissue at a time when required to a bioassembly head. The singularisation system consisted of a fluidic block fabricated from polycarbonate. The fluidic block was designed so that the hydrodynamic forces in the block could be varied sequentially using hydraulic valves and a pinch valve to both manipulate and trap micro-tissues and to achieve singularisation (figure 3).
Figure 3. Fluidics-based micro-tissue singularisation strategy. (a) Agitation in the chamber to clear any blockages that might be present, (b) capture port capturing the leading micro-tissue (c) flush back of the lagging micro-tissues and (d) the release and exit of the leading micro-tissue downstream. The arrows show the direction of fluid flow and the dotted arrow indicates the direction of pressure. (H) hopper valve, (F1) flush valve 1, (F2) flush valve 2, (P) pinch valve, (CP) capture port pressure valve and (CV) capture port vacuum valve. Dotted box = valve open, solid box = valve closed. (e) photograph of the singularisation chamber, with the leading micro-tissue captured and the lagging micro-tissues trailing behind.
Download figure:
Standard image High-resolution imageThe block consisted of three small vertical pressure ports and a horizontal main chamber. The arrangement of chambers is illustrated in figure 3. The vertical pressure ports—flush port 1, flush port 2 and capture port—are connected to pressurised reservoirs, with the fluidic pressure in the ports controlled by the four fluidic valves—hopper valve (H), flush valve 1 (F1), flush valve 2 (F2), capture port pressure valve (CP) and capture port vacuum valve (CV).
The main fluidic chamber was designed with an internal diameter of 1.3 mm to accommodate micro-tissues (or any other spherical aggregate or bioassembly tissue unit) with a diameter of 1 mm [23] for initial proof of concept, and also prevented horizontal stacking of the micro-tissues. The pressure ports were designed with a reduced diameter of 0.7 mm, in order to restrict micro-tissue entry into the ports and to be able to capture or trap the micro-tissues in position. The distance between the pressure ports on the main chamber was designed to be minimal to reduce the presence of a dead volume where micro-tissues may become trapped. A pressure sensor at the capture port measured the pressure as a feedback loop to identify successful micro-tissue capture or if blockages were present. Furthermore, a photomicrosensor downstream of the singularisation system was used to provide feedback if successful singularisation (i.e. the delivery of a single micro-tissue to the injection system) had occurred or not.
A summary of the singularisation process is illustrated in figure 3, and can primarily be broken down into 4 main steps—agitation, capture of leading micro-tissue, clearing of micro-tissue and release of leading micro-tissue. Micro-tissues entered the horizontal main chamber from the hopper reservoir which had a pneumatic hopper valve (H) to let air in the tank. Expulsion of micro-tissues from the singularisation device was controlled by a pinch valve (P). Pneumatic valves and pressure sensors were used to regulate the pressurised reservoirs containing media. Control software (LabVIEW, National Instruments, USA) and user interface was designed which took inputs from pressure sensors, the control system processes and feedback, user inputs as well as also sequentially controlling valve timings. Preliminary tests using formalin fixed micro-tissues were used to determine the optimal valve timing and pressure settings for optimisation of the singularisation system.
2.8. Design of the injection system
For the integrated tissue assembly device, a delivery subsystem was also required to automate the delivery of micro-tissues via two bottom-up fabrication strategies: (i) two-step bioassembly of micro-tissues at target locations within a prefabricated scaffold with defined pore spacing, or (ii) multistep bioassembly of micro-tissues at a target location via alternating layer-by-layer scaffold fabrication and assembly approach. To achieve this, an injection system was designed to successfully achieve both of these flexible bioassembly strategies and to minimise damage to the micro-tissues during the assembly process.
Figure 4 describes the steps involved in the injection concept in detail and also demonstrates the process for successful injection of Ø1 mm micro-tissues within the 3D plotted scaffold. Briefly, the assembly process begins with accurate positioning of the injection nozzle over the centre of the scaffold pore using the Bioscaffolder tool path calculated via the programming code (G-code) used to fabricate the scaffold. A solenoid operated expanding rod located within the core of the injection nozzle was then retracted to make way for the micro-tissue. The movement of the micro-tissue through the injection system was fluidically controlled, again by triggering the appropriate valves with specific control over valve timing via a LabVIEW software interface (see figure 7). Along with fluid flow, the downward movement of the expanding rod (figure 4(e)) aided the press-fit delivery of the micro-tissue into the pore. Furthermore, the expanding rod further helped to maintain a smooth profile for the nozzle so that the nozzle did not damage the scaffold fibres during the injection process and also protected the delicate nozzle tip during the placement of the nozzle on the scaffold fibre.
Figure 4. Micro-tissue injection strategy. (a) The injection system is positioned above the scaffold pore spacing where the micro-tissue is to be delivered, (b) it is then vertically lowered so that the expanding rod and nozzle sit on top of the upper fibres, (c) the expanding rod is retracted enabling (d) the delivery of the micro-tissue to the pore, (e) the expanding rod is lowered and pushes the micro-tissue in place if necessary and (f) the injection system is raised about the scaffold and is ready for the next insertion. The injection process. (g) Positioning the injection head, (h) micro-tissue singularisation and rod retraction and (i) micro-tissue insertion.
Download figure:
Standard image High-resolution image2.9. The integrated tissue assembly system
The integrated platform for the fabrication of 3D plotted scaffolds and automated assembly of micro-tissues consisted of the 3D Bioscaffolder and LabVIEW controlled singularisation and injection device. The 3D Bioscaffolder allowed conventional scaffold fabrication via fibre deposition but also serves as a 3D positioning system for the micro-tissue injection head (figure 5) which was mounted on the printer head with the use of a tool changer interface. The transport of single micro-tissues from the singularisation system to the injection system under fluid flow was achieved through a coupling tube.
Figure 5. Photograph of the micro-tissue injection head with labels showing the position of (i) hopper tank (where micro-tissues are loaded), (ii) pressure sensor and circuitry, (iii) Bioscaffolder 3D positioning head with tool changer, (iv) hydraulic valves, (v) singularisation chamber, (vi) pinch valve, (vii) injection solenoid, (viii) photosensor, and (ix) Bioscaffolder base plate.
Download figure:
Standard image High-resolution imageFor construct biofabrication, a scaffold layer (0°–90°–90°–0° fibre orientation with desired fibre spacing) was 3D plotted with a high temperature thermoplastic polymer print head, after which a micro-tissue was singularised and precisely assembled into the desired location within the scaffold via the injection system. The multistep process was repeated to create a bioassembled hybrid construct.
2.10. Micro-tissue shape characterisation pre and post-singularisation
Micro-tissue (HNC) dimensions (n = 9) were analysed both before and after undergoing the singularisation process. Samples were fixed overnight with 4% neutral buffered formalin, and imaged using a bright-field microscope. The major and minor diameters were measured using the image measurement function in Micrometrics SE Premium 4 software. Following imaging and measurement, micro-tissues were put through the singularisation device and then imaged and measured again as above, for comparison.
2.11. Reliability and efficiency of the singularisation device
Preliminary tests with the singularisation device demonstrated that HNC micro-tissues with a major diameter less than 0.82 ± 0.06 mm (n = 3) became lodged into the pressure ports (diameter of 0.7 mm) of the fluidic chamber, whereas micro-tissues that had a major diameter greater than 1.26 ± 0.02 mm (n = 3) did not enter the fluidic chamber (diameter of 1.3 mm). Micro-tissues with a major diameter in between this range could be handled by the singularisation device. To determine the optimal size range for reliable and efficient operation of the device and determine the efficiency of the singularisation device at the upper or lower limits of the micro-tissue diameter, the fabricated micro-tissues were fixed and sorted into two size ranges—group A and group B (see table 1).
Micro-tissues belonging to group A were then singularised in sets of 6 and repeated 17 times whereas micro-tissues belonging to group B were singularised in sets of 9 and repeated 12 times, with percentage success rate for singularisation recorded. The total number of successful singularisation cycles was used to determine the overall efficiency of the singularisation system (equation (4)). The number of successful singularisation cycles detected was used to determine photomicrosensor detection efficiency (equation (5)).
Table 1. Average major and minor diameter of HNC micro-tissues.
| Average diameter | ||
|---|---|---|
| Major | Minor | |
| Group A (n = 6) | 1.14 ± 0.12 mm | 1.03 ± 0.18 mm |
| Group B (n = 9) | 0.95 ± 0.07 mm | 0.84 ± 0.06 mm |
2.12. Viability of micro-tissues
In order to determine if either of the automated fluidic-based steps for singularisation, transport and injection influenced cell viability, HNC micro-tissues were divided into 4 groups, a control group (i.e. not put through the device), a singularised group (i.e. put through the singularisation device only), an injected group (i.e. put through the injection device only) and a singularised and injected group (i.e. put through both the singularisation and injection devices). The viability of the micro-tissues were determined using live/dead assay and the trypan blue exclusion assay.
For the live/dead assay, samples were incubated at 37 °C in 0.5 ml of Dulbecco's phosphate-buffered saline (DPBS; Invitrogen, USA) with 1 μM Calcein AM (Molecular Probes, USA) for 15 min, then 1.5 μM Propidium Iodide (Molecular Probes, USA) was added and incubated for 10 further minutes. Samples were then washed twice with DPBS and a z-stack of the sample was imaged using a Zeiss Axioimager Z1 microscope (FITC and Texas Red filter-set).
For the trypan blue exclusion assay, micro-tissue samples were digested overnight with 0.15% (w/v) collagenase type II (Worthington, USA) in basic chondrocyte media as described previously [23]. The cells were then diluted in an equal amount of 0.04% trypan blue (Gibco, USA) and the viability of the cells was quantified by imaging and counting of stained cells using a Zeiss Axioimager Z1 microscope.
2.13. Automated micro-tissue bioassembly
To validate the process for automated bioassembly of a 3D construct, a bilayered scaffold was designed and 3D plotted to incorporate 16 HAC micro-tissues in each layer adopting a layer-by-layer scaffold fabrication and micro-tissue bioassembly approach. For the first layer of the construct, 8 fibre layers were plotted and subsequently 16 viable micro-tissues were singularised and inserted using the automated assembly system. This process was repeated for the second layer.
During the micro-tissue insertion process, the volume of media flowing through the nozzle (figure 4(d)) was sufficient to keep the micro-tissues within the construct hydrated, and the surface tension ensured that the construct remained immersed in media. However, for the 3D plotting of the subsequent layer of the scaffold, the media level around the construct was manually adjusted with a pipette so that the fluid level remained below the uppermost scaffold fibre. This approach ensured that the resident micro-tissues remained hydrated (and viable) whilst also allowing the subsequent 3D plotted fibre to correctly solidify and adhere to underlying scaffold layers without interference of any media. An automated media level control system is currently under development.
2.14. Efficiency of micro-tissue insertion
A bilayered scaffold (n = 6) was assembled with 4 HAC micro-tissues in each layer using the layer-by-layer approach described above. The efficiency of micro-tissue insertion into the scaffold was determined by the ratio of successful micro-tissues inserted into the target scaffold pore to the total attempted number of micro-tissues inserted with the automated system (equation (6)).
2.15. Determination of viability of assembled construct
The layer-by-layer approach based bilayered scaffold (n = 3) with 4 viable HAC micro-tissues per layer was assembled using the automated system. Furthermore, a manually assembled scaffold (n = 3) was prepared as a control to compare against bioassembled constructs. In the latter case, the scaffold was 3D plotted and micro-tissues were inserted manually into prefabricated scaffold pores using a pipette fitted with a 1 ml pipette tip.
Live/dead assay was performed on manual and automated assembled constructs as described for micro-tissues above. To quantify metabolic activity in bioassembled constructs an AlamarBlue® assay was performed. AlamarBlue® (Invitrogen, USA) was added to the serum free basic chondrocyte media to a final concentration of 10% (v/v), and the samples were incubated at 37 °C for 3.5 h. The reduction in AlamarBlue® reagent was quantified colorimetrically by measuring the absorbance at 570 nm, using 600 nm as a reference wavelength (Fluostar Galaxy BMG Labtechnology, Germany).
2.16. Automated bioassembly of complex hierarchical hybrid construct
The proof of principal for bioassembly a complex hierarchical hybrid constructs with the automated bioassembly system was investigated by fabricating a hemispherical biphasic construct consisting of Gelatin-methacryloyl (GelMA) and methacrylated heparin (HepMA) micro-spheres. GelMA and HepMA were synthesised based on methods described previously [47, 56]. The fabricated hemispherical biphasic construct was representative of an osteochondral construct targeting automated hybrid bioassembly strategies for joint resurfacing. GelMA bioink micro-spheres were prepared by employing a microfluidic oil-emulsion setup based on previous work by Serra et al [56]. A visible light photo-polymerisation system previously developed in our group [57] was used for high throughput fabrication of 1 mm diameter micro-spheres consisting of either 10 wt% GelMA or 9.5 wt% GelMA supplemented with 0.5 wt% HepMA, and 2 mM/2 mM Ru/SPS photoinitiator (ruthenium/sodium persulfate; Sigma-Aldrich, USA). Micros-pheres were photo-crosslinked via radical polymerisation upon exposure to visible light (400–450 nm, 100 mW cm−2). Fabricated GelMA micro-spheres were stained with safranin-O (Sigma-Aldrich, USA) while GelMA-HepMA micro-spheres were stained with Coomassie brilliant blue (Thermo Fisher Scientific, USA) for 10 min and then washed 3 times in PBS to depict a biphasic bone phase (GelMA) and cartilage phase (GelMA-HepMA).
A computer aided design (CAD) model of a hemisphere with a radius of 10 mm was created in SOLIDWORKS and an STL file generated. The 3D plotting parameters and pore architecture described earlier in section 2.1 were used to fabricate the scaffold. The hemispherical biphasic construct was bioassembled via the layer-by-layer bottom-up approach. Firstly, 8 layers of fibres were 3D plotted with the high temperature plotting head and then micro-spheres were inserted utilising the micro-tissue injection head. For the subsequent layers, 4 layers of fibres were 3D plotted and the micro-spheres bioassembled for each particular layer alternating between hierarchical red (Safranin-O) and blue (Coomassie brilliant blue) labelled micro-spheres. The layer-by-layer fabrication process was repeated until the entire hemispherical construct was assembled. The red and blue labelled micro-spheres were used to represent the chondrogenic and osteogenic GelMA bioink regions of the hemispherical osteochondral construct.
2.17. Histology and immunohistochemistry
Bioassembled micro-tissues and cell-laden GelMA/GelMA-HepMA hydrogel micro-spheres were collected over 28 or 35 day culture in serum free chondrogenic differentiation media followed by fixation in 4% formaldehyde for 1 h at room temperature. Micro-tissue samples were dehydrated in a graded ethanol series, cleared in xylene, embedded in paraffin wax and sectioned into 5 μm thick slices. Micro-sphere samples were instead embedded in optimal cutting temperature compound (O.C.T) after fixation and cryo-sectioned into 30 μm thick slices, as per previously described protocols [23, 47]. Haematoxylin (Merck Millipore, NZ), Safranin-O and fast green staining (Sigma-Aldrich, MO) was applied to visualise cell nuclei, extracellular glycosaminoglycan (GAG) and collagen. Collagen type II was stained by immunohistochemistry using the R.T.U Vectastain Universal ABC kit (Vector Laboratories, USA) and the ImmPACT DAB peroxidase substrate (HRP; Vector Laboratories) according to the manufacturer's description. In brief, antigens were retrieved by treatment with pronase (1 mg ml−1, Sigma-Aldrich, MO) and hyaluronidase (10 mg ml−1, Sigma-Aldrich, MO) followed by incubation with canine and murine primary antibodies for collagen type I (1:300, ab34710; Abcam, Australia) and collagen type II (1:100, II-II6B3; Developmental Studies Hybridoma Bank) and secondary biotinylated equine antibodies for mouse and rabbit Ig (1:50, BA-1400 Vector Laboratories). Sections were further counterstained with haematoxylin to visualise the cell nuclei. GAG deposition in samples containing co-polymerised HepMA was visualised using aggrecan specific immunohistological techniques as heparin stains positive with Safranin-O [47]. In brief, antigen epitopes were retrieved by incubation in 0.1% hyaluronidase and constructs were blocked with 2% bovine serum albumin (BSA). Primary antibodies for aggrecan were diluted (1:200, Abcam, Australia) in blocking buffer and applied overnight at 4 °C followed by incubation with goat-anti-mouse (Alexa Fluor® 488, 1:400, Thermo Fisher Scientific, NZ) secondary antibodies. Constructs were further incubated with blocking buffer containing 4',6-Diamidino-2-Phenylindole, Dihydrochloride (DAPI, 1:1000, Thermo Fisher Scientific, NZ), as per previous described protocol [47]. All images were captured using a Zeiss Axioimager Z1 microscope.
2.18. Statistical analysis
Data was presented graphically as mean ± standard deviation. Data were analysed using GraphPad Prism v.6.0. Statistical analysis was performed using 1-way ANOVA or paired t-test, with p < 0.05 set as criterion for statistical significance.
3. Results
3.1. Scaffold characterisation
SEM images (figure 6) of the 3D plotted scaffold showed regular interconnecting pores in the in the x–y plane and z plane for the specific scaffold architecture and 3D plotting parameters adopted.
Figure 6. Scanning electron microscopy (SEM) images (x50) of the top view and sectional front view of the 3D plotted scaffold.
Download figure:
Standard image High-resolution imageThe theoretical and measured values of interconnecting pore size in the x–y plane and z-plane, the volume percent (Vol% porosityscaffold) porosity and the fibre diameter of the 3D plotted scaffold is indicated in table 2. The theoretical values for interconnecting pore size, Vol% porosityscaffold and fibre diameter were comparable with the measured values, where the average interconnecting pore size in the x–y plane was specifically designed at 780 μm to allow a slight press-fit of the Ø1 mm micro-tissues. It was essential to design the scaffold in this manner so that the micro-tissues could reliably be inserted into the pore by the automated bioassembly system, while also at the same time ensuring they remained securely in place and were not dislodged after the insertion process.
Table 2. Characterisation of 3D plotted scaffold.
| Interconnecting pore size | ||||
|---|---|---|---|---|
| x–y plane (μm) | z plane (μm) | Vol% porosityscaffold | Fibre diameter (μm) | |
| Theoretical | 780 | 440 | 84.17 | 220 |
| Measureda | 767.02 ± 16.54 | 427.25 ± 9.78 | 78.17 ± 2.26 | 218.50 ± 7.94 |
a(n = 4).
It is well-established that mechanical properties of 3D plotted scaffolds can be influenced by their architecture [2, 58]. For the particular architecture used in this study where the fibres were 3D plotted in a 0–90°–90°–0° pattern, the unconfined dynamic stiffness of the empty scaffold was determined to be 6.91 ±0.62 MPa at 1 Hz. For articular cartilage, 1 Hz is the range of physiological frequency representative of light to moderate activity [59]. In comparison, the dynamic stiffness of bovine cartilage under cyclic compression at 1 Hz has been reported to be approximately 9.6 MPa [58], whereas for bovine carpometacarpal cartilage a dynamic stiffness of 7.0 MPa at 1 Hz of cyclic compression has been reported [60]. The dynamic stiffness for human articular femoral cartilage at 1 Hz has been reported to be 4.5 MPa [61]. Therefore our data suggest that the dynamic stiffness of the scaffold design adopted for bioassembly in this study are similar to that of articular cartilage (at 1 Hz).
3.2. Timing of the singularisation system
Initially a prototype singularisation and injection system was designed and fabricated. After which, we examined and determined the most optimal valve and fluid flow timing sequences for the system to allow successful automated assembly. Figure 7 shows the timing diagram of the valve sequence for one singularisation cycle. This particular valve sequence timing was specific for a 10 cm long fluidic tube connecting the singularisation chamber and the hopper tank, a positive pressure of 105.7 kPa in the positive pressure tank, a negative pressure of 93.5 kPa in the negative pressure tank. A feedback control system assumed that whenever the pressure at the capture port had decreased to a value less than 96.5 kPa, a micro-tissue had been captured at the port, signalling to the control system to change the valve sequence from capturing the leading micro-tissue to clearing lagging micro-tissues. Varying any of the above parameters would require a recalibration of the valve timing sequence. This sequence formed the initial framework for the operation of the device. The time required to singularise and inject a micro-tissue within a pore of the 3D plotted scaffold ranged from 8 to 15 s.
Figure 7. Timing diagram of the valve sequence for a single cycle of singularisation. For valve positions see figure 3(H) hopper valve, (F1) flush valve 1, (CP) capture port pressure valve (CV), capture port vacuum valve, (F2) flush valve 2 and (P) pinch valve.
Download figure:
Standard image High-resolution image3.3. Micro-tissue analysis pre and post-singularisation
To understand and scrutinise any physical deformation and mechanical impact on micro-tissues after being passed through the singularisation system, HNC micro-tissues were imaged and measured pre and post-singularisation. The average percentage change in the major and minor diameter of micro-tissues (n = 9) measured pre and post-singularisation is listed in table 3. The values demonstrated that there was no significant change in micro-tissue size and shape (p > 0.05) as a result of being handled by the device.
Table 3. Average diameter of HNC micro-tissues pre and post-singularisation and percentage change in diameter post-singularisation in paired samples.
| Average diameter | |||
|---|---|---|---|
| Pre-singularisation | Post-singularisation | Percentage change in diameter post-singularisationa | |
| Major diameter | 1.04 ± 0.16 mm | 1.03 ± 0.16 mm | 2.2 ± 2.8% |
| Minor diameter | 0.99 ± 0.17 mm | 0.98 ± 0.15 mm | 2.7 ± 3.4% |
3.4. Reliability and efficiency of the singularisation device
The reliability of the device was quantified by determining the efficiency of successful micro-tissue (HNC) singularisation. As demonstrated in table 4, the singularisation efficiency for micro-tissues of larger sizes (group A) was 97.0 ± 6.6%, whereas it was 86.0 ± 13.8% for the smaller micro-tissue group (group B). This result indicates that micro-tissues with a diameter closer to the upper limit of the device have better singularisation efficiency as long as they are not large enough to obstruct and cause a blockage in the singularisation chamber. Successful operation of the tissue bioassembly system therefore depends on the ability to fabricate tightly controlled regular-sized spherical micro-tissues and our group has previously described a high throughout approach demonstrating this [23].
Table 4. Efficiency of singularisation.
| Average diameter | |||
|---|---|---|---|
| Major | Minor | Singularisation efficiencya | |
| Group A | 1.14 ± 0.12 mm | 1.03 ± 0.18 mm | 97.0 ± 6.6% |
| Group B | 0.95 ± 0.07 mm | 0.84 ± 0.06 mm | 86.0 ± 13.8% |
During the experiment, the situations which resulted in singularisation failures included: (i) capture of 2 micro-tissue at a time, (ii) failure to trigger the capture pressure sensor although a micro-tissue had been captured, (iii) accidental release of micro-tissue upstream rather than downstream, (iv) blocking of pressure ports by the micro-tissues, and (v) accidental dislodging of the captured micro-tissue. Nevertheless, the determined efficiency of ∼97% for the targeted (group A) micro-tissues was deemed to be acceptable for its application in tissue bioassembly. The singularisation failures that occurred were not critical and did not cause the failure of the device nor the inability to completely assemble all available micro-tissues. For example, in the event of a failure, the singularisation cycle could be repeated if the device had failed to release a micro-tissue, or in the case of a blockage, the device could be purged with fluid so as to dislodge the blocked micro-tissue and clear the bioassembly system after which normal operation was resumed. In the case of these failures however, the overall bioassembly time for the entire construct would increase slightly.
The reliability of the photomicrosensor in detecting a micro-tissue was based on the number of successful detection of micro-tissue singularisation and was found to be 96.0 ± 5.2%.
3.5. Viability of micro-tissues
Significant attention was invested in designing the bioassembly system to ensure that the environment within the device was favourable for cell survival and that, size or shape of the micro-tissues was not adversely affected. For that reason, a fully fluidic approach was adopted for singularisation and delivery of the micro-tissues. To ascertain whether the cells in the micro-tissues were affected during their passage through the device, a live/dead assay was carried out to inspect cell viability on the surface of the HNC micro-tissues. In addition, a trypan blue exclusion assay was performed to quantify cell viability throughout the entire HNC micro-tissue.
Although cells at the surface of micro-tissues were most prone to mechanical or physical deformation during the singularisation and injection process, live/dead analysis (figure 8) indicated that cells were predominantly viable and unaffected by the process. This was further validated by trypan blue exclusion assay (figure 9) indicating no significant difference (p > 0.05) in percentage of viable cells between the control (80.45 ± 4.77%, non-singularised/non-injected micro-tissue) and either singularised (83.45 ± 3.43%) or injected (78.38 ± 2.37%) or both singularised and injected (83.55 ± 5.53%) micro-tissues. This data suggests that the automated bioassembly system presented in this study does not impart adverse effects on micro-tissues.
Figure 8. Fluorescence microscopy images of (a) control (b) singularised (c) injected and (d) singularised and injected HNC micro-tissues stained with Calcein AM (live cells, green) and Propidium Iodide (dead cells, red).
Download figure:
Standard image High-resolution imageFigure 9. Percentage of viable cells determined using the Trypan blue exclusion assay for control, singularised, injected, and singularised and injected HNC micro-tissues. The data is presented as mean ± SD (n = 4). No significant difference (p > 0.05).
Download figure:
Standard image High-resolution image3.6. Automated tissue bioassembly
The ability of the automated tissue bioassembly system to assemble a hybrid construct was demonstrated following the validation of the individual bioassembly device components.
A scaffold consisting of 1 mm fibre spacing was 3D plotted with the high temperature plotting head as described above, and HAC micro-tissues were inserted into the pores using the micro-tissue injection head (see figure 5). The time required to fabricate the construct utilising the automated bioassembly system was approximately 20–25 min. To improve visualisation and to differentiate engineered cartilage tissue modules from surrounding scaffold, the micro-tissues were stained with Safranin-O. The steps involved in the layer-by-layer 3D plotting and micro-tissue assembly of a bilayered construct with the aid of the automated bioassembly system is as shown in figure 10.
Figure 10. (a)–(h) Top view and sectional front view photographs of the steps involved in the layer-by-layer plotting and assembly of a bioassembled construct consisting of high throughout fabricated HAC micro-tissues (0.25 × 106 cells/micro-tissue) with the aid of the automated system. The construct contains 32 micro-tissues, arranged in 2 layers with 16 micro-tissues in each layer. (i) Higher magnification image of the assembled construct. The micro-tissues were stained with Safranin-O for visualisation.
Download figure:
Standard image High-resolution image3.7. Efficiency of micro-tissue insertion
The efficiency of successful micro-tissue (HAC) insertion into the scaffold was determined to be 79.2 ± 18.8%. Although there were micro-tissues that failed to be inserted into the scaffold, the scaffold was visually inspected by the user for errors at the end of assembling every layer and the micro-tissue insertion process was repeated for pores not containing a micro-tissue, thereby allowing the complete scaffold bioassembly as designed.
3.8. Viability of bioassembled constructs
After demonstrating that the fully fluidic approach did not damage micro-tissues, we needed to ascertain that the micro-tissues were not affected by the automated system inserting the micro-tissues within the designated pores of the plotted scaffold. Although the dispensing temperature in the high temperature print head was approximately 200°, the extruded material rapidly cools to ambient temperature and solidifies. Therefore, during the layer-by-layer plotting process, we had to ensure that the molten polymer being plotted on the previous layer of assembled micro-tissues (HAC) did not affect cell viability.
Visual inspection of the live/dead fluorescence microscopy images of the manually assembled construct (i.e. micro-tissues were press-fit by hand into a 3D plotted scaffold) and the construct assembled using the bioassembly system showed no obvious differences (figure 11). A localised reduction in cell viability was however observed at the periphery of micro-tissues adjoining the scaffold in both manually assembled and automated assembled constructs. This observation could be caused by the shear forces experienced by the cells on the surface of the micro-tissues during the micro-tissue insertion process, a well-recognised phenomena within the field of biofabrication [62–65]. It should be noted that there was no significant difference (p > 0.05) in metabolic activity (figure 12) between manually assembled constructs (62 ± 4%) and constructs assembled with the automated bioassembly system (58 ± 7%). These results indicated that it is unlikely that the high temperature of extruded PEGT/PBT fibres laid down during the subsequent scaffold layer fabrication during bioassembly was responsible for any reduction in cell viability given that manual assembly of micro-tissue constructs occurred at room temperature.
Figure 11. Fluorescence microscopy images of (a) manually assembled construct and (b) construct assembled using the bioassembly system stained with Calcein AM (live cells, green) and Propidium Iodide (dead cells, red).
Download figure:
Standard image High-resolution image3.9. Automated bioassembly of complex hierarchical hybrid construct
The potential of the automated bioassembly system to fabricate a complex hierarchical hybrid construct was demonstrated by successfully fabricating a hemispherical biphasic construct with GelMA micro-spheres (figure 13(b)) as an osteochondral biphasic model for joint resurfacing. The bioassembled construct demonstrates the feasibility of the automated bioassembly system to accurately fabricate large hybrid constructs with predetermined architecture and mechanical stability. Bioassembled chondrocyte micro-tissues and chondrocyte-laden GelMA-HepMA micro-sphere constructs were further cultured in serum free chondrogenic differentiation media for 28 and 35 days, respectively, to validate the compatibility of both the high-throughput fabrication of micro-tissues and the automated bioassembly system during long-term tissue culture. A gradual increase of both GAG and collagen type II staining was observed out to 28 days in bioassembled micro-tissues (figures 14(a)–(f)), as established markers of hyaline cartilage [38, 66], consistent with our previous reports of high-throughput micro-tissue fabrication [23, 67]. The histological sections further highlight that the fusion of adjacent micro-tissues over time supports the secretion of ECM components without undifferentiated or necrotic cells forming in the central regions. Bioassembled chondrocyte-laden GelMA-HepMA micro-spheres further demonstrated the capability of the automated bioassembly platform to fabricate complex hierarchical constructs while supporting both long-term cell viability (figure 14(g)) and deposition of cartilage-specific, GAG protein aggrecan (figure 14(h)) out to 35 days. These results further showcase the versatility of the developed bioassembly platform, with demonstrated capability to facilitate direct spatial organisation and hierarchical 3D assembly of micro-tissue modules, ranging from biomaterial free cell pellets to cell-laden hydrogel formulations.
Figure 12. Percentage of AlamarBlue® reduced to determine cell viability in constructs fabricated via manual bioassembly and automated bioassembly approaches. The data is presented as mean ± SD (n = 3) and no significant difference was observed (p > 0.05).
Download figure:
Standard image High-resolution imageFigure 13. (a) Illustration of a computer aided design (CAD) example of an assembled hemispherical construct for osteochondral joint resurfacing. (b) A biphasic hemispherical construct with stained GelMA hydrogel micro-spheres representing chondrogenic (red) and osteogenic (blue) phase of an osteochondral construct fabricated by applying the bottom-up automated tissue bioassembly strategy. Scale bar = 2 mm.
Download figure:
Standard image High-resolution imageFigure 14. Sections of assembled micro-tissues and associated tissue fusion in adjacent culture over 28 days (a)–(f) stained with safranin-O/haematoxylin/fast green (a)–(c) or Collagen II antibodies (e), (f). Bioassembled HAC-laden 9.5% GelMA-0.5% HepMA micro-spheres (g), (h) stained with Calcein AM (live cells, green) and Propidium Iodide (dead cells, red) (g) or DAPI (blue) and Aggrecan (purple) antibodies (h) after 35 days culture in chondrogenic differentiation media.
Download figure:
Standard image High-resolution image4. Discussion
Typically, traditional tissue engineering strategies utilise a 'top-down' approach, where cells are seeded into a prefabricated biomaterial scaffold to create an engineered tissue. However, such 'top-down' approaches with their inability to precisely control the spatial distribution of cells and support material have difficulty recreating the intricate microstructural features of tissues [1]. In contrast, 'bottom-up' biofabrication or modular tissue engineering approaches focus on fabricating engineered tissue by the self-assembly or directed-assembly of a tissue unit from smaller components, or modules, with specific micro-architectural features [7, 31]. Furthermore, as many tissues are comprised of repeating functional units, there is a strong biological basis for using biofabrication and/or bottom-up approaches to generate complex hierarchical and functional engineered tissue constructs [1].
Modular tissue engineering aims to create biomimetic structures by designing modular tissues that can be used as building blocks to create larger tissues [1]. These modules can be created via a number of approaches, such as self-assembled aggregation [68], micro-fabrication of cell-laden hydrogels [69], creation of cell sheets [70], direct printing of cells [71] or via high throughput pellet or micro-mass culture techniques [23] as described herein. By successfully testing and establishing a reliable and efficient blueprint for automated manipulation of micro-tissue 'modules' combined with 3D plotting of structural 3D scaffolds, potentially overcomes barriers to building large and complex high quality living constructs of clinically relevant sizes, with precision, in large numbers, and with a reduced construction time.
Automated biofabrication technology platforms ensure that tissues based on mathematical or anatomical models generated through non-invasive imaging techniques such as computed tomography (CT) or magnetic resonance imaging (MRI) can easily be translated into a computer-aided design and computer aided manufacturing (CAD/CAM) file and input into 3D bioprinting devices [72]. The application of computer-aided technologies has led to the development of a new biofabrication platforms and more recently the convergence of multiple top-down and bottom-up bioprinting strategies with cell-laden bioinks to generate hybrid constructs [73, 74]. We believe that we have paved a path that employs computer aided technology to create scaffolds with a wide array of properties and geometries. The bioassembly strategy and system described herein offers a submodule of a computer-controlled tooling process for assembling complex hybrid 3D tissue models in a high throughput and repeatable manner for screening as well as anatomically matched, hierarchical tissue constructs for regenerative medicine.
The described fluidic-based system offers advantages over other methods such as laser based writing or jetting techniques where the cells experience extreme kinematic profile during the cell transfer process [75–77]. Cell-based constructs fabricated using cell direct writing techniques also have a downfall in that they lack an initial support structure and the maturation of the bioactive tissue must be rapid so as to have the necessary structural support [76]. From this study, the absence of any detrimental effect on viability and shape of micro-tissues after singularisation and bioassembly reflects the gentle nature of the designed fluidic system.
We have previously described a tightly controlled regular-sized spherical micro-tissue fabrication process in a high throughput fashion [23, 31]. The ability to fabricate micro-tissues with controlled size is critical as micro-tissues closer to the upper limit of the device have improved singularisation efficiency and thereby a lower rate of errors during construct bioassembly. Although the described proof of concept system works efficiently with micro-tissue diameters that are within the designed size range of Ø1 mm, the system has an innate shortcoming in that micro-tissues above or below this designed size range cannot be efficiently handled. However, by suitably modifying the diameter of the singularisation chamber channels and the injection nozzle and expanding rod, the bioassembly system can be adopted for micro-tissues of other desired sizes (for example <500 μm). The required optimal micro-tissue or hydrogel micro-sphere size potentially compatible with the device can be obtained by adjusting cell seeding densities during micro-tissue fabrication process as previously demonstrated [37, 39, 67], or by modifying the flowrate during microfluidic fabrication of cell-laden micro-spheres [78]. We have generated micro-tissues with human chondrocytes and human MSC ranging from Ø250 μm to Ø1.5 mm utilising the high throughput micro-tissue fabrication technique described in this paper.
Importantly, the tissue bioassembly system can potentially be used to assemble micro-tissues or micro-spheres of different cell types in a specific order by either utilising (i) a single singularisation device loaded with the desired micro-tissue, after which other subsequent micro-tissue types can be loaded and assembled for a particular layer, or (ii) multiple parallel singularisation devices loaded with different types of micro-tissues, respectively, can be rigged up to a common injection device used for the assembly process. The use of the technology in this fashion would have important implications in co-culture systems where two or more cell types are brought together in the same culture environment either to enhance tissue formation of a specific lineage and/or to form a multicellular tissue or organ replacement [50]. In cartilage engineering, this can be applied to chondrocytes and synovial fibroblasts [50], chondrocytes and osteoblasts [50], primary and passaged chondrocytes [79], and MSC and chondrocyte [80, 81] co-cultures.
We demonstrated that automated bioassembly could be used to assemble a simple hybrid construct where a complete scaffold is prefabricated using 3D plotting, and micro-tissues are assembled into the scaffold. Alternatively, a more complex multistep bottom-up approach involving layer-by-layer fabrication where the scaffold is 3D plotted and micro-tissues are co-assembled to generate complex hierarchical constructs was also demonstrated. The impact of this would be the ability to create anatomically shaped complex hybrid constructs (figure 13). Furthermore, bioassembly of pre-differentiated micro-tissues as well as chondrocyte-laden GelMA-HepMA hydrogel micro-spheres demonstrated the capability of the platform to fabricate complex hierarchical constructs while supporting tissue fusion, long-term cell viability, and deposition of cartilage-specific ECM proteins (GAG, aggrecan) out to 28 or 35 days in vitro (figure 14). These results showcase the versatility of bioassembly as an alternative biofabrication platform, with demonstrated capability to accurately assemble both biomaterial free micro-tissue and cell-laden hydrogel micro-sphere modules to fabricate designed, hierarchical 3D hybrid constructs of clinically relevant shape and size.
The bioassembly system could be applied in micro-tissue applications in other fields of biofabrication. Assembled cancer micro-tissues or spheroids may play a valuable role in translational research in cancer biology and drug testing by recreating the natural physical and structural environment of living tissues, and the use of a tissue assembly system is an attractive choice [82]. However, cancer spheroids tend to have a large variation in aggregation pattern between different cell types and this would potentially require modification of the system to accommodate the assembly of certain types of cancer spheroids [83]. In high throughput drug screening (HTS), the use of micro-tissues as a 3D cell culture system offers an improved model of in vivo behaviour of most cell types, as opposed to monolayer or suspension cultures [84]. HTS is another area where the system could be adopted to handle and manipulate micro-tissues.
5. Conclusions
We described the development of an automated and integrated biofabrication system for 3D bioassembly of micro-tissues within structural 3D plotted scaffolds. The singularisation device effectively delivered single micro-tissues to the injection print head which—with the aid of the 3D positioning system—facilitated accurate delivery of individual micro-tissues to desired locations within a 3D plotted scaffold. We demonstrated that the fluidic-based bioassembly system efficiently handled individual micro-tissue, with no detrimental effect or damage to cells or micro-tissue modules as part of the automated singularisation and injection bioassembly phases. No difference in cell viability was observed between constructs containing manually assembled micro-tissues and those constructs fabricated with the automated bioassembly system. The developed system was capable of fabricating simple hybrid constructs via a two-step bottom-up bioassembly strategy involving 3D plotting of a complete scaffold and injection of micro-tissues, or generating complex hierarchical hybrid constructs via a multistep bottom-up bioassembly strategy combining layer-by-layer scaffold fabrication and micro-tissue co-assembly. This technology paves the way for adoption of automated biofabrication systems to produce hierarchical bioassembled tissues or co-culture models for high-throughput screening or complex and mechanically stable hybrid tissue constructs of clinically relevant size.
Acknowledgments
We acknowledge funding from the Royal Society of New Zealand Rutherford Discovery Fellowship (TW, RDF-UOO1204) and the AO Foundation (S-08-81W). We would like to acknowledge Professor Paul Dalton, Jonathon Parrish, Julian Murphy, Stefanie Sperling, Ben Jar and Jeremy Nicholls for their scientific discussions and technical input to the manuscript.